This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison.
Slides for my Final Presentation: |
| ||||||
Conclusions
Prader-Willi Syndrome affects about 1 in 15,000 individuals worldwide and is the number one genetic cause of obesity in children. It is characterized most by a loss of muscle tone in the infant and an increase in fat between the ages of 1 and 6. Prader-Willi individuals suffer from the inability to feel full after eating. As you might imagine, this alone causes behavioral problems and may contribute to the obesity phenotype. The disorder is also characterized by cognitive disabilities, abnormal facial characteristics, and incomplete sexual development (1). Prader-Willi is caused by a deletion of the 15th chromosome, resulting in the deletion of 5 genes, depending on the person (2). One of these genes is necdin, whose gene products play a major role in neural development and cell cycle regulation, among other functions (3). An integral part to this 321 amino acid long protein is the MAGE domain, a domain that is a part of upwards of 25 other proteins, playing a role in either tumor suppression or cell differentiation. In necdin protein, the MAGE domain plays a role especially in cell differentiation through DNA binding and protein binding within the nucleus and cytoplasm (4). Necdin, itself, is very well conserved, though it is found only among mammals, so it is relatively newly evolved. Among mammals, necdin ranges from 96% identity in the rhesus macaque to a 81% identity in the mouse. Because this conservation is so high and mice have been used often in studying Prader-Willi syndrome, I will be using mice throughout my specific aim proposals.
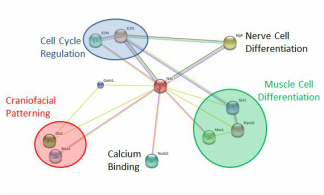
Before getting further into my aims, it is important to look at the protein interactions already found with necdin and what their functions are, as this gives us an idea of what necdin could play a role in besides what is already known. As seen in the figure to the left, necdin's interaction partners include proteins involved in cell cycle regulation, nerve cell differentiation, muscle cell differentiation, calcium binding, and craniofacial patterning (5).
Because I was especially interested in necdin's muscle cell differentiation partners, I looked further into this. I also had questions about what necdin's role in the obesity phenotype of PWS. I found an article that helped me start to piece these ideas together.
It is known that necdin plays a role in the upregulation of muscle differentiation and a downregulation of fat differentiation (6). This led me to questioning whether the loss of necdin in Prader-Willi plays a role in the transition between low muscle tone in infants and an increase in fat later in child development. In looking through more information about PWS, I found a knowledge gap in what happens to fat and muscle cell differentiation during development of Prader-Willi Syndrome. My hypothesis, based on my prior research, is that the loss of necdin decreases the amount of muscle differentiation and increases the amount of fat differentiation at a certain point in development. I believe that this point of development will be at the same age stage that the transition between low muscle tone and increased fat occurs in in PWS patients, between age 1 and 6.
Before getting further into my aims, it is important to look at the protein interactions already found with necdin and what their functions are, as this gives us an idea of what necdin could play a role in besides what is already known. As seen in the figure to the left, necdin's interaction partners include proteins involved in cell cycle regulation, nerve cell differentiation, muscle cell differentiation, calcium binding, and craniofacial patterning (5).
Because I was especially interested in necdin's muscle cell differentiation partners, I looked further into this. I also had questions about what necdin's role in the obesity phenotype of PWS. I found an article that helped me start to piece these ideas together.
It is known that necdin plays a role in the upregulation of muscle differentiation and a downregulation of fat differentiation (6). This led me to questioning whether the loss of necdin in Prader-Willi plays a role in the transition between low muscle tone in infants and an increase in fat later in child development. In looking through more information about PWS, I found a knowledge gap in what happens to fat and muscle cell differentiation during development of Prader-Willi Syndrome. My hypothesis, based on my prior research, is that the loss of necdin decreases the amount of muscle differentiation and increases the amount of fat differentiation at a certain point in development. I believe that this point of development will be at the same age stage that the transition between low muscle tone and increased fat occurs in in PWS patients, between age 1 and 6.
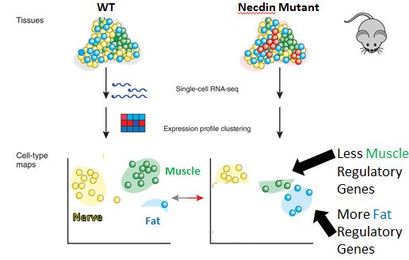
Aim 1: Are more fat cell associated genes and less muscle cell associated genes found in Necdin mutants?
In my first study, I want to first see whether there is a correlation between more fat and less muscle with a loss of necdin. To accomplish this, I can use an RNA-seq approach to determine amounts of fat vs. muscle regulatory genes. By gaining this information, we will be able to infer the amount of fat and muscle cells formed in relation to each other. My hypothesis is that there will be a gain in fat regulatory genes and a loss of both muscle and nerve regulatory genes in the necdin mutant compared to the WT mice.
In my first study, I want to first see whether there is a correlation between more fat and less muscle with a loss of necdin. To accomplish this, I can use an RNA-seq approach to determine amounts of fat vs. muscle regulatory genes. By gaining this information, we will be able to infer the amount of fat and muscle cells formed in relation to each other. My hypothesis is that there will be a gain in fat regulatory genes and a loss of both muscle and nerve regulatory genes in the necdin mutant compared to the WT mice.
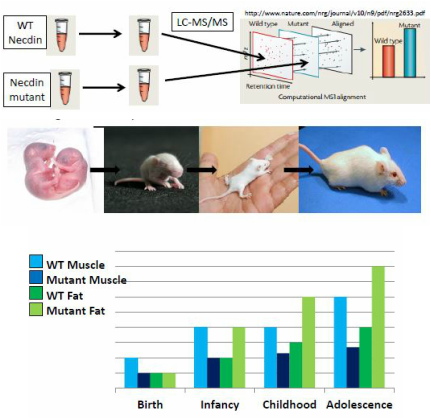
Aim 2: Is there overall muscle cell loss and fat cell accumulation at a certain stage in development with loss of necdin?
My next step is to determine necdin's role throughout development in fat cell and muscle cell differentiation. To do this, I will use a quantitative mass spectrometry approach. We will take tissue from both WT and necdin mutant mice from different stages in development (birth, infancy, early childhood, and adolescence). Before thinking about mutant phenotypes, we must first know the normal development of both fat and muscle. Muscle growth occurs at high rates in early infancy, plateaus in childhood, and increases again with puberty. Fat, on the other hand, develops at a slow rate throughout development. With this knowledge combined with knowledge of PWS phenotypes, lead us to the hypothesis that with loss of necdin, muscle will have decreased growth right away at birth accompanied with slow growth throughout development. Fat development will begin normally but then increase exponentially between infancy and early childhood. When translated into protein quantities found in mass spectrometry, there will be increased fat regulatory proteins in each stage after infancy and decreased muscle regulatory proteins right away at birth, in comparison to WT mice regulatory proteins. This information will give us information about the timescale of PWS phenotypes so as to develop a timescale for diagnosis.
My next step is to determine necdin's role throughout development in fat cell and muscle cell differentiation. To do this, I will use a quantitative mass spectrometry approach. We will take tissue from both WT and necdin mutant mice from different stages in development (birth, infancy, early childhood, and adolescence). Before thinking about mutant phenotypes, we must first know the normal development of both fat and muscle. Muscle growth occurs at high rates in early infancy, plateaus in childhood, and increases again with puberty. Fat, on the other hand, develops at a slow rate throughout development. With this knowledge combined with knowledge of PWS phenotypes, lead us to the hypothesis that with loss of necdin, muscle will have decreased growth right away at birth accompanied with slow growth throughout development. Fat development will begin normally but then increase exponentially between infancy and early childhood. When translated into protein quantities found in mass spectrometry, there will be increased fat regulatory proteins in each stage after infancy and decreased muscle regulatory proteins right away at birth, in comparison to WT mice regulatory proteins. This information will give us information about the timescale of PWS phenotypes so as to develop a timescale for diagnosis.
Aim 3: What are necdin protein interactions related to fat and muscle throughout development?
In my final aim, I want to determine further necdin protein interactions and their gene ontology so as to gain more information on necdin's role in fat and muscle differentiation processes at different stages of development. To do this, I will be taking necdin protein from the same stages of development as in Aim 2. With this protein, I will use a tap tag approach to determine more possible protein interaction partners than already known. The tap tag approach is a really efficient method to create a large protein interaction network. After finding new protein interaction partners, we will determine their gene ontology to see whether any function in muscle or fat development. With this study, I have two hypotheses. One, necdin will interact with more muscle regulatory proteins, but new fat regulatory proteins will be found. For example, one of the already known interaction partners, NGFR, may play a role in fat differentiation, and I am sure there are more in this network. Secondly, I believe that necdin will interact with different protein types at different stages of development. Therefore, not only will the protein interaction network expand with better screening methods, the network will change throughout different stages of development.
In my final aim, I want to determine further necdin protein interactions and their gene ontology so as to gain more information on necdin's role in fat and muscle differentiation processes at different stages of development. To do this, I will be taking necdin protein from the same stages of development as in Aim 2. With this protein, I will use a tap tag approach to determine more possible protein interaction partners than already known. The tap tag approach is a really efficient method to create a large protein interaction network. After finding new protein interaction partners, we will determine their gene ontology to see whether any function in muscle or fat development. With this study, I have two hypotheses. One, necdin will interact with more muscle regulatory proteins, but new fat regulatory proteins will be found. For example, one of the already known interaction partners, NGFR, may play a role in fat differentiation, and I am sure there are more in this network. Secondly, I believe that necdin will interact with different protein types at different stages of development. Therefore, not only will the protein interaction network expand with better screening methods, the network will change throughout different stages of development.
Future Directions:
After these experiments, there are many next steps to be taken. Among these, I think it will be important to determine further the relationship between NGFR, Necdin, and fat differentiation as mentioned in Aim 3. Also, we can gain a lot of information about necdin's functioning by further looking into each of the new protein interaction relationships found in Aim 3. Next, I want to create a diagnosis plan using the timeline information found in Aim 2, as earlier diagnosis of the disorder can lead to a better prognosis for children suffering from PWS. Finally, i want to more generally explore the relationship of necdin in fat and muscle cell differentiation throughout development further. Not much information is yet known about this relationship, so any studies that can be done to look further in this will help push forward PWS research.
After these experiments, there are many next steps to be taken. Among these, I think it will be important to determine further the relationship between NGFR, Necdin, and fat differentiation as mentioned in Aim 3. Also, we can gain a lot of information about necdin's functioning by further looking into each of the new protein interaction relationships found in Aim 3. Next, I want to create a diagnosis plan using the timeline information found in Aim 2, as earlier diagnosis of the disorder can lead to a better prognosis for children suffering from PWS. Finally, i want to more generally explore the relationship of necdin in fat and muscle cell differentiation throughout development further. Not much information is yet known about this relationship, so any studies that can be done to look further in this will help push forward PWS research.
References
1. Basic Facts About PWS. (n.d.). Retrieved April 6, 2015, from http://www.pwsausa.org/about- pws/basic-facts-about-pws
2. Genes and mapped phenotypes. (n.d.). Retrieved May 16, 2015, from http://www.ncbi.nlm.nih.gov/gene/4692
3. Gene Ontology Consortium. (n.d.). Retrieved May 16, 2015, from http://geneontology.org/
4. Necdin. (n.d.). Retrieved May 16, 2015, from http://www.uniprot.org/uniprot/Q99608
5. STRING: Functional protein association networks. (n.d.). Retrieved May 16, 2015, from http://string-db.org
6. Bush, J., & Wevrick, R. (2012). Loss of the Prader–Willi obesity syndrome protein necdin promotes adipogenesis. Gene, 45-51
2. Genes and mapped phenotypes. (n.d.). Retrieved May 16, 2015, from http://www.ncbi.nlm.nih.gov/gene/4692
3. Gene Ontology Consortium. (n.d.). Retrieved May 16, 2015, from http://geneontology.org/
4. Necdin. (n.d.). Retrieved May 16, 2015, from http://www.uniprot.org/uniprot/Q99608
5. STRING: Functional protein association networks. (n.d.). Retrieved May 16, 2015, from http://string-db.org
6. Bush, J., & Wevrick, R. (2012). Loss of the Prader–Willi obesity syndrome protein necdin promotes adipogenesis. Gene, 45-51
|
| ||||||||||||
